Dr. Poonam
Hello! My name is Dr. Poonam, and I am from India. I have completed my Bachelor Degree in Combined Sciences, Master in Environmental Biotechnology and Doctorate in Biofuel Production From M.D University, India. I have working experience of 5 and half years with School and University students. The year ahead will be probably the most important of your son or daughters academic career so far, and I see my role as supporting them at this crucial time to achieve of their very best, in close collaboration with their form tutors, with the teaching staff and with you as parents. I look forward to improving the education of your child!
This year we will have some fun and challenging tasks to help us reach our goals. We are going to work, play, and learn hard and have fun in the process. Through our daily interaction, we will learn and prove there is no "I" in "TEAM." Our class goal will be to work together towards a fantastic and successful school year! I have high expectations for all of my students and I am committed to assisting them with the expectations required of the course.
Best Regards,
Dr.Poonam
Hello! My name is Dr. Poonam, and I am from India. I have completed my Bachelor Degree in Combined Sciences, Master in Environmental Biotechnology and Doctorate in Biofuel Production From M.D University, India. I have working experience of 5 and half years with School and University students. The year ahead will be probably the most important of your son or daughters academic career so far, and I see my role as supporting them at this crucial time to achieve of their very best, in close collaboration with their form tutors, with the teaching staff and with you as parents. I look forward to improving the education of your child!
This year we will have some fun and challenging tasks to help us reach our goals. We are going to work, play, and learn hard and have fun in the process. Through our daily interaction, we will learn and prove there is no "I" in "TEAM." Our class goal will be to work together towards a fantastic and successful school year! I have high expectations for all of my students and I am committed to assisting them with the expectations required of the course.
Best Regards,
Dr.Poonam
Term-1 Syallbus
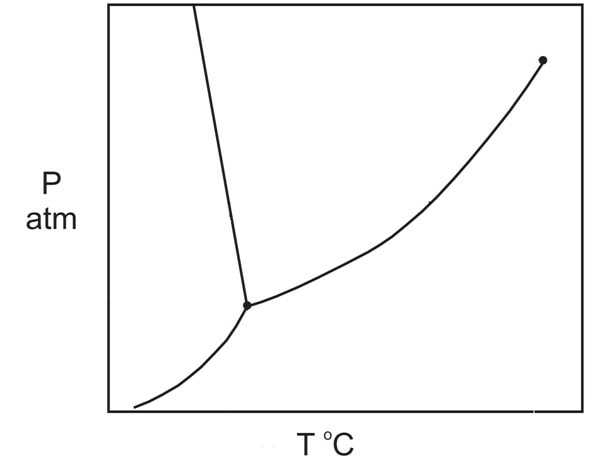
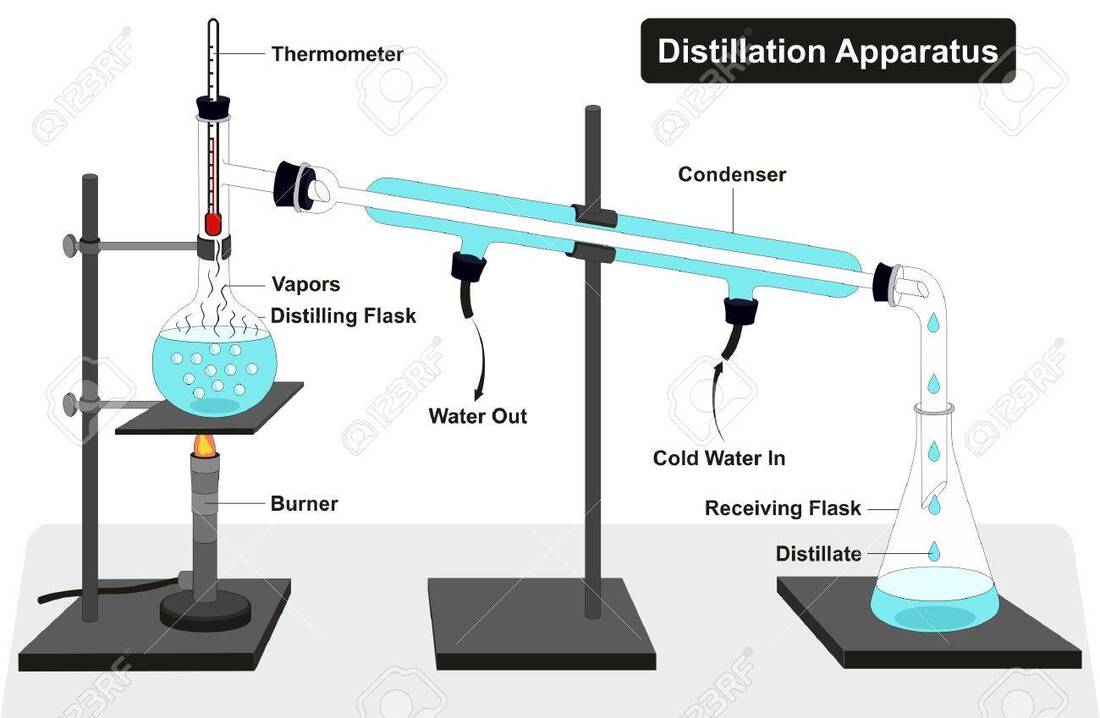
CHAPTER 1. THE PARTICULATE NATURE OF MATTER
CHAPTER 2. ELEMENT, COMPOUND AND EXPERIMENTAL TECHNIQUES
CHAPTER 3. ATOMIC STRUCTURE AND BONDING
CHAPTER 1. THE PARTICULATE NATURE OF MATTER
Chapter-2 Element, compound and experimental techniques
Quiz On element, compound and mixtures
Mid-Term Syallbus
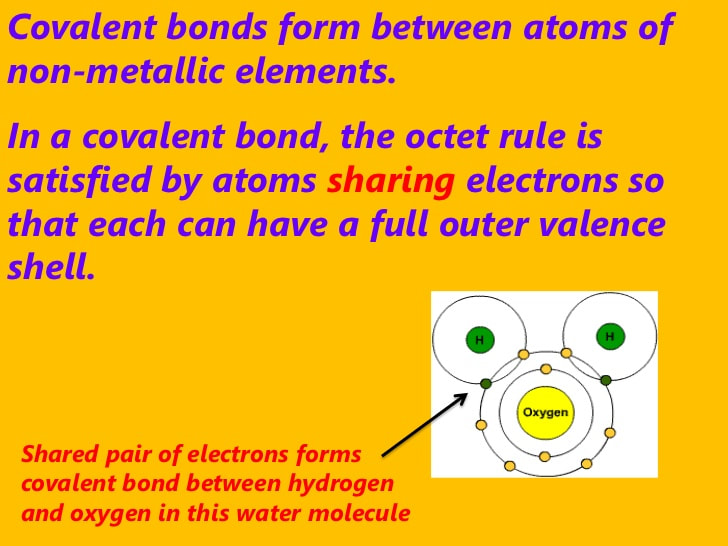
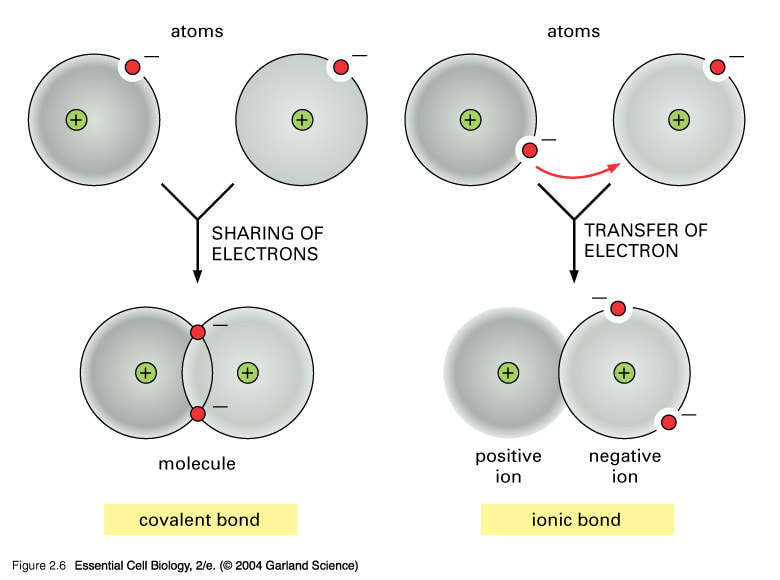
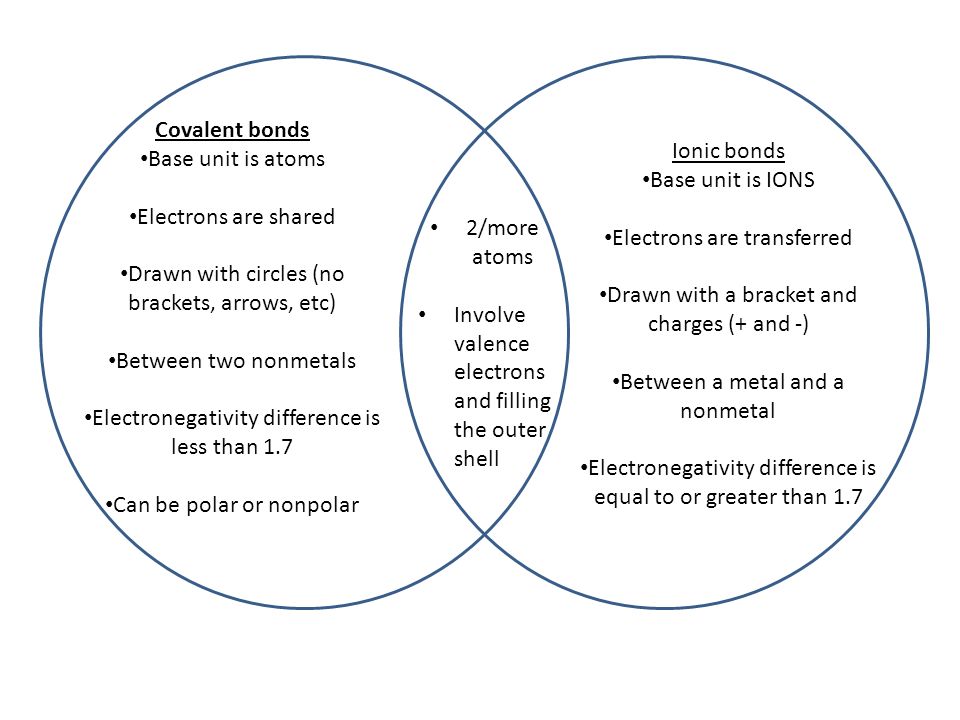
Chapter-3 Atomic structure and bonding
Chapter-3 Atomic structure and bonding
Term- 2
Chapter-5 Electricity and Chemistry
Holidays Homework
Review the Following topics:
1. The Particulate nature of matter
2. Element, compounds and mixtures
3. Atomic structure and bonding
4. Electricity and chemistry
5. Solve the following sample Papers (Download the pdf file from below)
1. The Particulate nature of matter
2. Element, compounds and mixtures
3. Atomic structure and bonding
4. Electricity and chemistry
5. Solve the following sample Papers (Download the pdf file from below)
Chapter-6 Chemical Energetics
Chapter-7. Chemical Reaction
Chapter 4- Stoichiometry- Chemical Calculation
Chapter-8 Acids, Bases and Salts
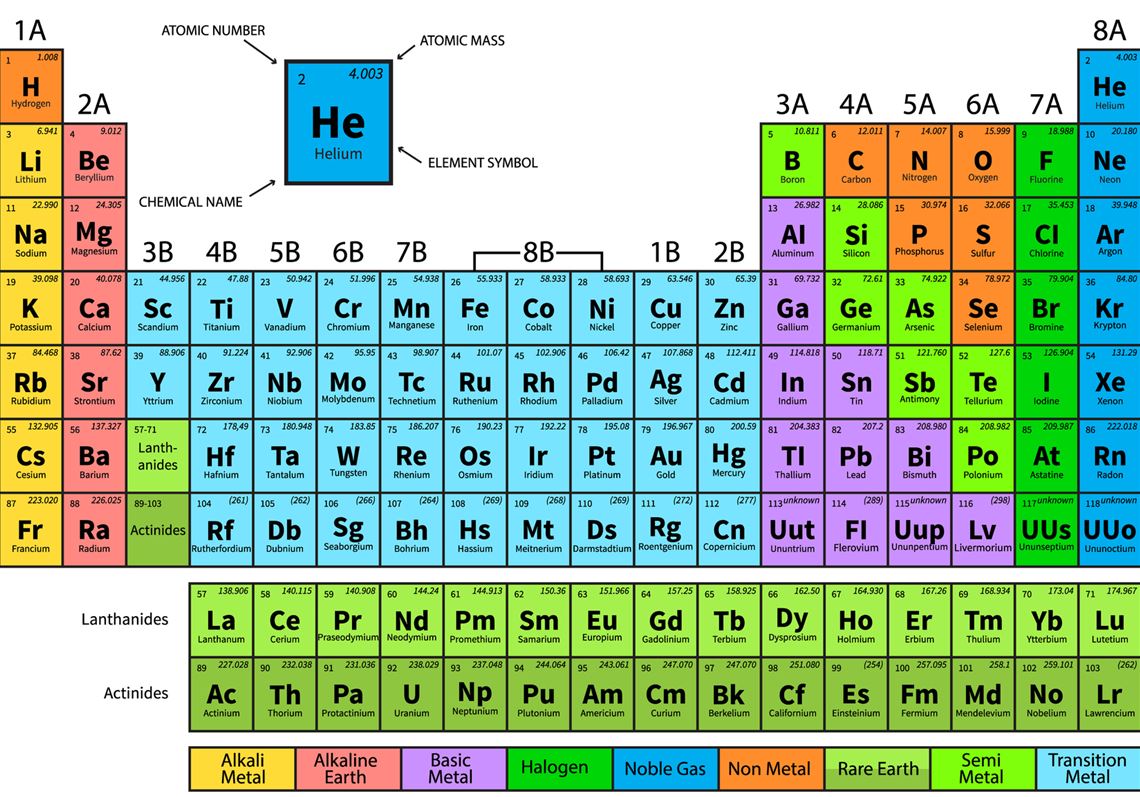
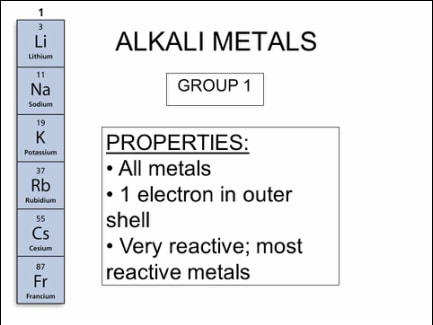
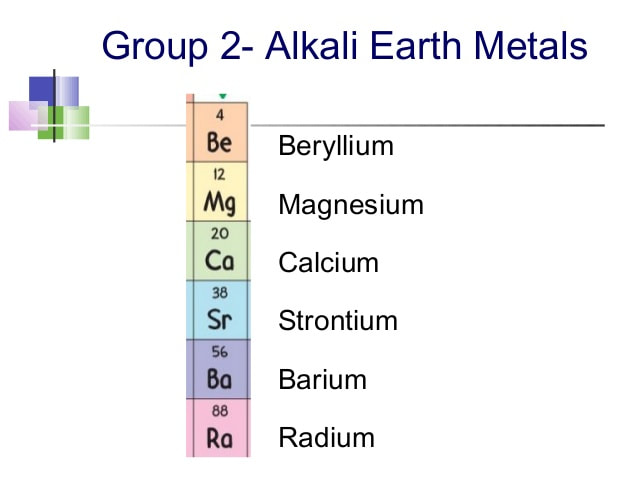
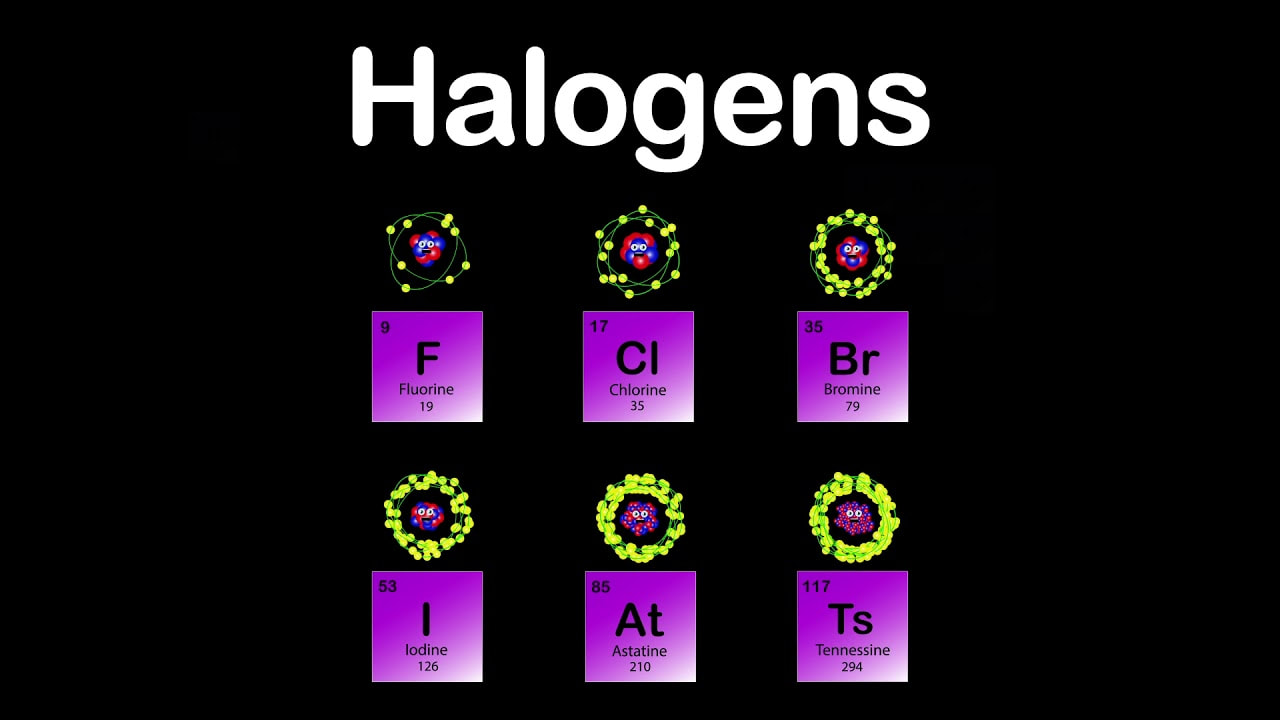
Chapter 9- The periodic table
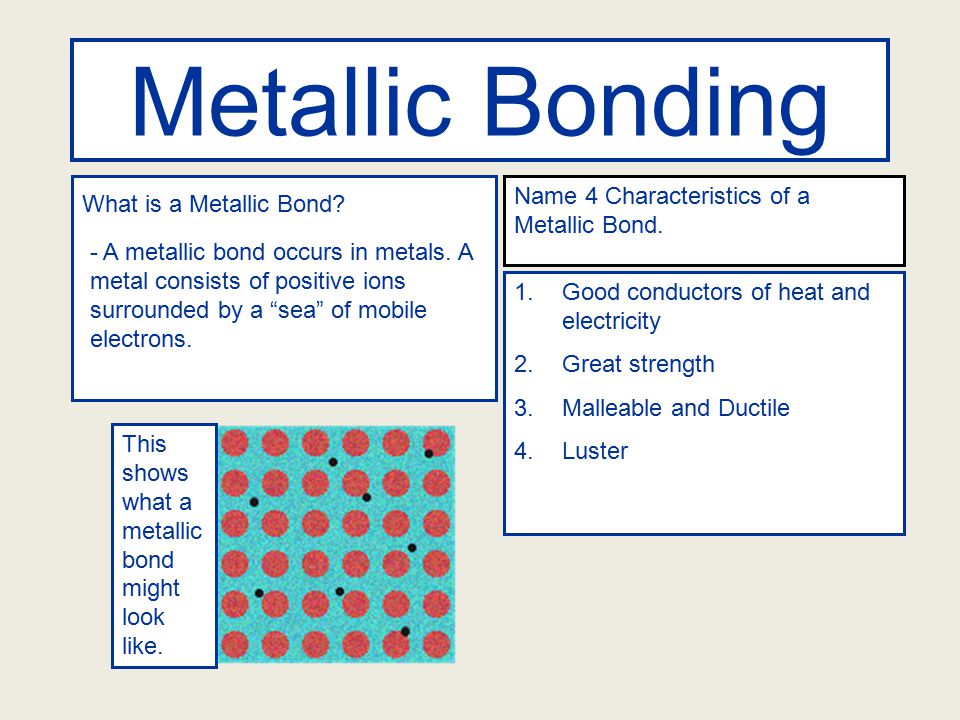
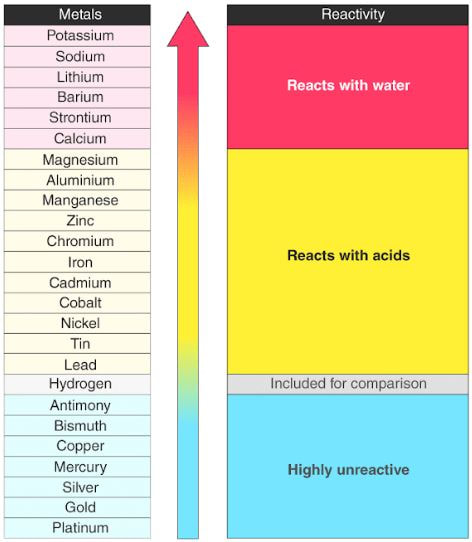
Chapter 10- Metals
Metals reactions
Extraction of metal
Some metals such as Potassium(K) and Sodium(Na) are very reactive rare in naturally pure form. Both of these metals are also difficult to extract.
Chapter 12 Sulfur
Sulfur Properties
Yellow and non-metallic
group VI on periodic table
Brittle
non conductive
Low melting point- 115 degrees celsiusC
Non water soluble
How we get Sulfur
Found in metal ores like copper pyrite (CuFeS2) Zinc blende (ZnS)
Found near volcanoes.
Found in natural gas and oil wells.
Uses of Sulfur
Make sulfuric acid- BATTERY ACID
Vulcanize rubber- makes rubber harder and more elastic
Vulcanize rubber- makes rubber harder and more elastic
Chemical properties of Sulfur
Reacts with both metals and nonmetals
Mg(solid) + S(solid)→ MgS(solid) S(solid) + O2 (gas)→ SO2(gas)
magnesium + sulfur→ magnesium sulfide sulfur + oxygen→ sulfur dioxide
Sulfur dioxide
Colourless gas produced when sulfur is burned
Mainly comes from vehicle exhaust-car/boat/plane
Choking smell and extremely poisonous- acidic SO2(g) + H2O(l) ⇋ H2SO3(aq)
Contributes to acid rain
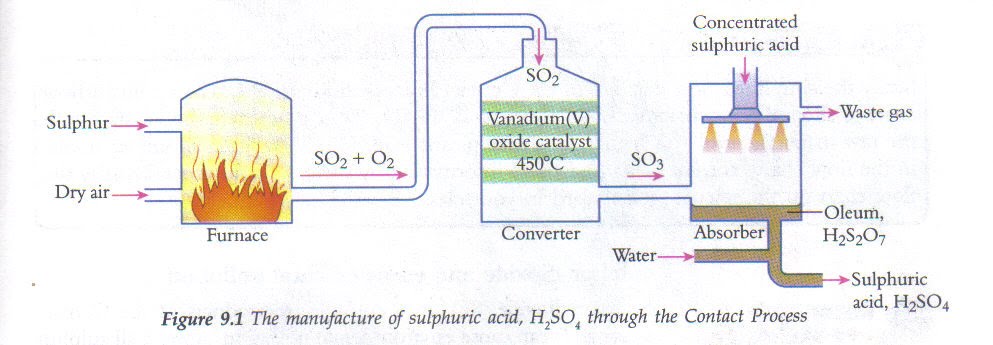
The Contact process
S(solid) + O2 (gas)→ SO2(gas) 2SO2(gas) + O2 (gas) ⇋ 2SO3
Burn sulfur to make sulfur dioxide. Pass that smoke over acatalyst (vanadium oxide) to make sulfur trioxide. Add water slowly to make sulfuric acid.
SO3 + H2O → H2SO4
Burn sulfur to make sulfur dioxide. Pass that smoke over acatalyst (vanadium oxide) to make sulfur trioxide. Add water slowly to make sulfuric acid.
SO3 + H2O → H2SO4
Sulfur as a Dehydrating agent
Concentrated Sulfuric acid can be used to pull water from substances
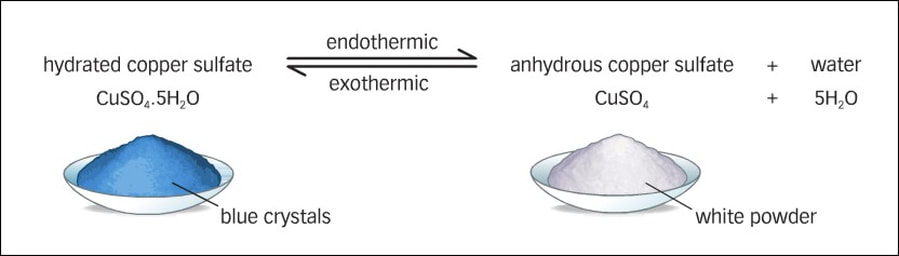
Ex: Cu12SO4⦁H2O(s) → CuSO4(s) + 5H2O(l)
hydrated copper (II) sulfate becomes anhydrous copper (II) sulfate and water in the presence of sulfuric acid.
Ex: Cu12SO4⦁H2O(s) → CuSO4(s) + 5H2O(l)
hydrated copper (II) sulfate becomes anhydrous copper (II) sulfate and water in the presence of sulfuric acid.
Sulfur as an Oxidizing agent
Concentrated sulfuric acid is a powerful oxidizer. Will react with metals!
Cu(solid) + 2H2SO4(liquid) → CuSO4(solid) + 2H2O(liquid) + SO2(gas)
Mix copper and concentrated sulfuric acid to make Copper (II) sulfate, water and sulfur dioxide
Cu(solid) + 2H2SO4(liquid) → CuSO4(solid) + 2H2O(liquid) + SO2(gas)
Mix copper and concentrated sulfuric acid to make Copper (II) sulfate, water and sulfur dioxide
dilute sulfuric acid - Dibasic acid
2 replaceable hydrogen atoms
Can produce 2 types of salt: normal salts & acid salts
Produces sulfates by reacting with bases(alkali)
Can produce 2 types of salt: normal salts & acid salts
Produces sulfates by reacting with bases(alkali)
Sulfates
Salts of sulfuric acid
Used for fertilizer, x-ray studies and making medical casts
Testing for sulfate
Tested for by adding a few drops of dilute HCl and a few drops of Barium Chloride.
Negative: no white precipitate forms
Positive: white precipitate forms
Negative: no white precipitate forms
Positive: white precipitate forms
Bulk Chemicals
Chemicals made in large amounts for many uses
Ex: ammonia, sulfuric acid, ethanol
Ex: ammonia, sulfuric acid, ethanol